 
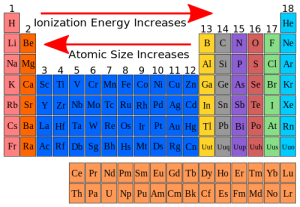
< < and < < Įlectronic configuration of the cation: for two cations of the same size and charge, the one with a pseudo noble-gas configuration (with 18 electrons in the outer-most shell) will be more polarizing than that with a noble gas configuration (with 8 electrons in the outermost shell). The cation charge increases (size decreases) and on the right, the anion size increases, both variations leading to an increase in the covalency. The greater the positive charge, the smaller the cation becomes and the ionic potential is a measure of the charge to radius ratio. Large cations are to be found on the bottom left of the periodic table and small anions on the top right.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |


 RSS Feed
RSS Feed
